- Amgen will invest $600M into a new science and innovation center in Thousand Oaks, California, breaking ground later this year.
- The facility will consolidate research and development functions but will not include drug manufacturing.
- The project underscores a broader trend in life sciences real estate, as pharmaceutical companies ramp up US-based operations to reduce dependency on global supply chains.
Back To Its Roots
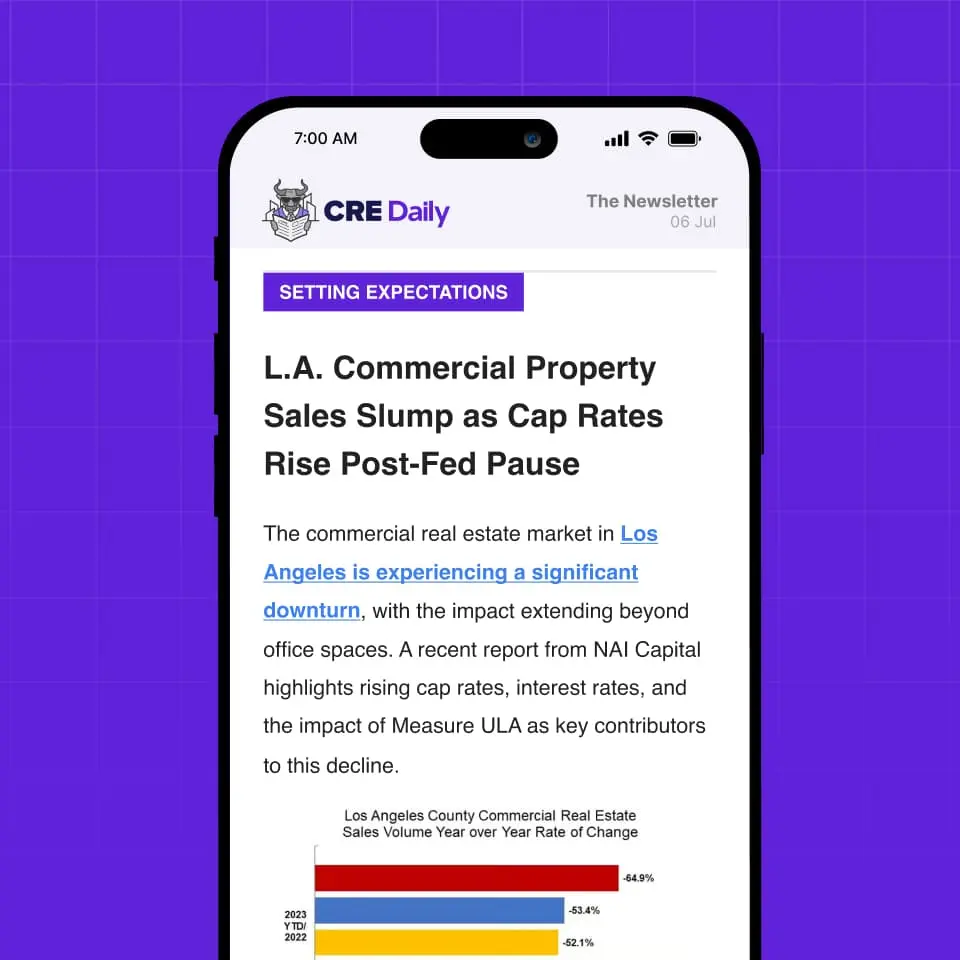
Amgen, one of the world’s largest biotech firms, is reaffirming its commitment to its Southern California base, reports CoStar. The company plans to begin construction in Q3 on a $600M science and innovation center in Thousand Oaks. It already operates close to 2M SF of biotech space in the area.
The new facility will serve as a central hub for scientists, engineers, and researchers. The center will not manufacture drugs. Instead, it is designed to accelerate the company’s R&D pipeline by bringing teams together in a collaborative setting.
Biotech Real Estate Rebound
The announcement comes amid a broader recovery in the life sciences real estate market, which struggled post-pandemic. Demand is surging, driven in part by pharmaceutical firms reshoring operations to guard against geopolitical risk, including tariffs and supply chain disruptions.
According to JLL, 15 major pharmaceutical companies have announced more than $270B in US-based projects this year—a 185% increase in biomanufacturing demand. Alexandria Real Estate Equities is the largest biotech landlord in the country. So far this year, it has added 500K SF of lab space. Another 1.1M SF is currently in the pipeline.
Get Smarter about what matters in CRE
Stay ahead of trends in commercial real estate with CRE Daily – the free newsletter delivering everything you need to start your day in just 5-minutes
Nationwide Expansion
Thousand Oaks isn’t Amgen’s only big real estate play. The company is also investing $900M to expand its production facility near Columbus, Ohio. In December, it committed $1B to build a new drug substance plant in Holly Springs, North Carolina.
Policy Tailwinds
Federal agencies are now moving to streamline the approval process for drug manufacturing sites. In August, the FDA launched a program to help speed up development timelines, some of which can stretch to a decade. The Biden administration has voiced support for these initiatives as part of a broader strategy to reduce US reliance on foreign pharmaceutical production.
Why It Matters
Amgen’s move reflects growing momentum in US-based life sciences development. As reshoring accelerates and government support increases, real estate tied to research and biotech manufacturing is gaining momentum. It stands out as a bright spot in an otherwise cautious commercial property market.
What’s Next
With strong demand and policy support, further biotech investment is expected to reshape suburban life sciences clusters across the US. This trend is especially evident in markets with existing infrastructure, such as California, North Carolina, and Ohio.